 On 24 September, 1987, six year old Leide Ferreira threw up ten minutes after eating her egg sandwich. The next day her parents started throwing up too. Vomiting and diarrhea were followed by strange aches and burns. When Leide’s mother Maria went to the public health clinic in Goiás, the doctor ascribed her symptoms to food poisoning and sent her back home. Leide’s grandmother, who came to help her family, got sick too. Lesions crept over their skin and their hair fell out. The family’s neighbors began to think they had AIDS.
On 24 September, 1987, six year old Leide Ferreira threw up ten minutes after eating her egg sandwich. The next day her parents started throwing up too. Vomiting and diarrhea were followed by strange aches and burns. When Leide’s mother Maria went to the public health clinic in Goiás, the doctor ascribed her symptoms to food poisoning and sent her back home. Leide’s grandmother, who came to help her family, got sick too. Lesions crept over their skin and their hair fell out. The family’s neighbors began to think they had AIDS.
On 28 September, Maria dragged herself back to the doctor’s office and deposited on his desk a small plastic bag filled with iridescent blue powder. This, she said, was the culprit. The doctor dismissed her claims as superstitious nonsense and admitted her to the Tropical Diseases hospital. But another doctor at the clinic called a health physicist to test the mysterious bag. The next day, 112 thousand people found themselves packed into Olympic stadium, queuing at hastily constructed tents to be tested for radiation poisoning.
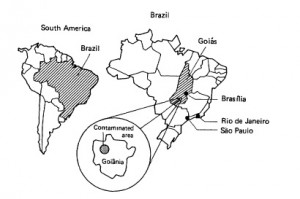 Goiânia is the capital of the state of Goiás in central Brazil. About 1.3 million people live there. In 1971, a local cancer treatment clinic bought a radiation therapy machine. When the practice closed 14 years later, the administrators left the now-obsolete machine in the abandoned clinic without notifying either the state or Brazil’s National Nuclear Energy Commission (CNEN).
Goiânia is the capital of the state of Goiás in central Brazil. About 1.3 million people live there. In 1971, a local cancer treatment clinic bought a radiation therapy machine. When the practice closed 14 years later, the administrators left the now-obsolete machine in the abandoned clinic without notifying either the state or Brazil’s National Nuclear Energy Commission (CNEN).
The deserted clinic went through the usual process of decomposition. By 1987, the building had been reduced to three ramshackle walls with large holes eating through the peeling sides. Homeless people slept there. The erstwhile treatment room contained human excrement and the still relatively intact radiological machine, which looks like a cross between an enormous telescope and a dentist’s chair.
That machine was finally discovered by Roberto dos Santos Alves and Wagner Mota Pereira, two locals who made their living selling scrap metal. On 13 September, the men brought a wheelbarrow to the dilapidated building, took apart the heavy outer assembly, and transported the inner canister back to Leide’s uncle’s scrap lot, where they got about $25 for it. When Devair Ferreira’s employees smashed the canister open, they found still another inner container. Ernesto, one of the employees, finally pried the capsule open with a screwdriver.
His curiosity was rewarded with about a third of an ounce of sparkling, glowing blue powder. The consistency was variable—some crystals were as big as rice grains; others the size of dust. They were held loosely together in a crumbly cake, which Ernesto broke apart and sifted through his fingers. Witnesses later said they remembered him calling it “carnival glitter.” Ernesto chose one of the bigger blue crystals to make into a ring for his wife, put it in his right front pocket, and went back to work.
The powder enthralled everyone. Devair began to suspect that it might be supernatural. His brother dipped a finger into the dust and drew a cross on his abdomen. Maria slept in bedclothes covered in the sparkly blue dust. Neighbors and acquaintances came to the Ferreiras’ home to take some of the mystical powder for good luck. Six-year-old Leide rubbed the powder all over her arms so that she glowed and sparkled. The bedclothes, the house, and the girl were covered in crystals. So was the egg sandwich she ate for lunch.
The blue glitter wasn’t magical, but it did have some powerful properties: the salt, called cesium chloride, was used in the radiotherapy machine. As its active ingredient, cesium-137, decays, it emits two radioactive products: beta particles and gamma rays, both of which damage the body. Beta particles are simply electrons. They travel slowly and without much force. At worst, external exposure irritates the skin, causing a sunburn-like reddening called “beta burn,” but skin cells are tough enough to easily block beta particles from doing any internal damage. But when they’re ingested, the story changes. The same sluggish travel capacity that makes them relatively harmless outside the body makes them more harmful inside, where all their energy is trapped inside a tiny radius. The more energy the electron burns into each cell, the worse the damage, and the more DNA strands are broken.
The other product of cesium-137 decay is gamma radiation. Gamma rays are photons, just like visible light, but with about 10,000 times as much energy. Gamma rays are more penetrating than beta particles, in fact more so than x-rays: they barely notice the skin as they glide through organs, muscle, and bone. Because they are so deeply penetrating, they may not hit any cells at all, but just pass through the body without interaction. Whether they hit a cell or pass through depends on how much radiation is emitted by the source and how close a person is to that source. The corollary of “beta burn” is called a thermal gamma burn, but instead of irritating the skin, it fries the organs inside the body.
The larger particles Leide had rubbed on her skin adhered to the sandwich and wreaked havoc on her stomach and intestine cells during digestion. The finest crystals clung to the dust she breathed and drifted into her lungs.
The time it takes to die of radiation poisoning depends on how the exposure compares to background radiation, the nominal amount of radiation a person can expect to receive from the natural environment. That’s around 0.002 Gray per hour, a Gray being the standard international unit of absorbed radiation dose. If gamma radiation can be thought of as a steady stream of progressively more lethal bullets, the Gray measures how many of those little bullets will hit during a given amount of time. A dose of 4-5 Grays—30,000 times the background levels—kills all the body’s red and white blood cells. The death of these cells destroys the immune system, and opportunistic diseases can roam free. The exposed individual dies within a few weeks.
 Once CNEN realized the magnitude of the contamination, the organization cast a wide net to test as many people as they could. They flew 54 severely contaminated people to a hospital in Rio de Janeiro, where they were put in “reverse isolation,” which, instead of protecting the staff from a patient’s infection, protects patients with compromised immune systems from infections caused by something as innocuous as a fungus or a common cold carried by a nurse’s aide. Hospital staff had to decontaminate patients’ skin anew every day, using a mix of soapy water and a diluted acid, because their sweat kept re-contaminating their own skin from the inside.
Once CNEN realized the magnitude of the contamination, the organization cast a wide net to test as many people as they could. They flew 54 severely contaminated people to a hospital in Rio de Janeiro, where they were put in “reverse isolation,” which, instead of protecting the staff from a patient’s infection, protects patients with compromised immune systems from infections caused by something as innocuous as a fungus or a common cold carried by a nurse’s aide. Hospital staff had to decontaminate patients’ skin anew every day, using a mix of soapy water and a diluted acid, because their sweat kept re-contaminating their own skin from the inside.
The first task, however, was to remove as much of the internal source as they could. “When you’re internally exposed, the first thing you want to do is just get it out of your body by any means necessary,” says Jonathan Links, a health physicist at Johns Hopkins University and the former president of the Nuclear Medicine Society. “You start with brute force: lavage, laxatives. Just get whatever is inside out.” In addition to the “brute force” tactics, they tried chemical methods. Prussian Blue is a chemical agent that works by bonding to the radioactive particles inside the body. These are then secreted through the normal pathways: urine, feces, and sweat.
Most of the 54 survived because they were exposed to less than 1 Gray. However, several people lost fingers, and 23 people were treated for localized radiation burns. Five required skin grafts. Ernesto, the scrap yard worker who had sifted lovingly through the entire cake of fairy dust, received intense thermal gamma burns on his hands and on his right thigh, where the cesium-137 chunk intended for his wife’s ring had burned through his trousers.
The men who had dismantled the cancer therapy machine in the clinic fared badly. According to the IAEA report detailing the incident [PDF], Pereira and Alves had probably been exposed to doses around 4 Gray from the moment they moved the giant machine. Both men had complained of vomiting, diarrhea and vertigo within days of their initial exposure, and Pereira’s hand was swollen and burned. Like Leide’s father, he had been initially diagnosed with a reaction to tainted food. Late in October, two of Devair’s other workers died of internal hemorrhaging.
For Leide and her mother, the only care the hospital could offer was palliative. Leide had an estimated internal dose of 4 Gray—that’s just from inhalation and ingestion—and she died on 23 October, on the same day as her mother. She was buried in a lead coffin, sealed with concrete.
Leide’s death was only the first tremor of what would happen in Goiânia. Less than half an ounce of cesium chloride would become a city-wide contamination, a political and economic catastrophe, and 13 and a half tons of nuclear waste. And that was just in the first month.
Last week, the New York Times reported that the reactors at the Fukushima Daiichi plant reactors have discharged mainly iodine-131 and cesium-137. “It’s the cesium that would prompt an evacuation,” Argonne National Laboratory nuclear engineer Shih-Yew Chen told Science, which also reported that Japan’s cesium-137 soil measurements are now surprisingly high.
What makes the Goiânia incident instructive is that unlike the Chernobyl disaster, which spewed everything—strontium, cesium, iodine and a whole assortment of nasty characters—here the sole contaminant was cesium-137. It has been possible to trace the effects, over 30 years, of a single source of a single radioactive isotope.
I’ll continue the story next time.
[The opening picture: It’s not cesium chloride. It’s art courtesy of flickr user bmooneyatwork. The map is courtesy of the 1988 International Atomic Energy Agency report on the incident. The third image shows people being tested for exposure at the stadium, courtesy of CNEN, and you can find it in the IAEA report. The fourth image is actual cesium, but not cesium chloride, by Wikimedia user Dennis “S.K.”]

This morning I started a post on my blog with:
Radiation is now being detected in Russia, imagine what it must be like for the poor Japanese who live within 500 miles. Japan is not that big of a place though it is a land of big people who have made some huge mistakes. Almost all supporters of the nuclear industry have run for cover but today we see one being sneaked out or savagely used.
“I think there is a role for safe nuclear power,” says Dr. David J. Brenner directs the Center for Radiological Research. He is the expert brought out by the New York Times to confuse everyone. Is there really such a thing as safe nuclear power? How anyone can say that after this latest nuclear accident is beyond comprehension.
I have posted almost every day since the start of this nuclear disaster….would like to include this post in my book coming out soon on Nuclear Toxicity Syndrome
Dr. Mark Sircus, Ac., OMD, DM(P)
@Mark: I think you’re coming at this from a very non-scientific angle. Nuclear power is, overall, safe. But, when we see a single instance like this, our minds trick us into thinking it’s a lot more likely than it actually is.
For the record, Fukushima really is a new situation. We’ve built plants to survive both earthquakes and flooding, but apparently not both at once. And, if the Fukushima situation is an object lesson in anything, it’s the fact that the power company running a plant can’t cut corners on safety procedures the way it was done there. Even with that, the vast majority of very bad outcomes were avoided by the countermeasures that were put in place.
When it comes down to it, nuclear power is “safe”. But, maybe the world itself just isn’t.
sloppy: “Science, which also reported that Japan’s cesium-137 soil measurements are now surprisingly high.”
All these readings were taken within a 50km radius. Japan is a larger country than that.
Is there a version of this story that has a left margin?
Mark Sircus,
Where is your doctorate from? I highly doubt it is accredited.
Also do more research. Yes nuclear is dangerous, but Coal is even more so, its just all of its problems are spread out over years as opposed to single accidents causing all the cases. In addition there are on the horizon Thorium reactors which are safer and more efficient than plutonium reactors, and can’t melt down. They are the future of safe nuclear power. You are just a fear monger trying to drum up business for yourself at the cost of the future.
Life is a series of risks of variable size. The question is, which risks are worth taking, which are not. People answer this question according to their values, their wants and needs, their emotions. Personally, I think the risks of nuclear power are worth taking as long as those responsible for it’s oversight and operation do their very to safeguard the rest of us – which they usually but not always have succeeded in doing.
“Dr. Sircus was trained in acupuncture and oriental medicine at the Institute of Traditional Medicine in Sante Fe, N.M., and at the School of Traditional Medicine of New England in Boston.
He served at the Central Public Hospital of Pochutla in Mexico, and was awarded the title of doctor of oriental medicine for his work. He was one of the first nationally certified acupuncturists in the United States.”
“His book, The Terror of Pediatric Medicine, is a free e-book offered on his web site.
…
He has recently released a number of e-books including Winning the War Against Cancer, Survival Medicine for the 21st Century, Sodium Bicarbonate, Rich Man’s Poor Man’s Cancer Treatment, New Paradigms in Diabetic Care and Bringing Back the Universal Medicine: IODINE.”
http://www.naturalnews.com/031974_radiation_dangers.html
“Sodium bicarbonate is the least expensive, safest and perhaps most effective cancer medicine there is. Sodium Bicarbonate is a nothing to lose everything to gain treatment for cancer.”
Rich Man’s Poor Man’s Cancer Treatment
http://publications.imva.info/index.php/e-books/sodium-bicarbonate-rich-man-s-poor-man-s-cancer-treatment-e-book.html
http://publications.imva.info/index.php/about-the-author
SIGNIFICANT MATH ERROR
Background radiation cited in the eleventh graf as 0.002 Gy/hr (2mGy/hr) is wrong, very wrong; off by three orders of magnitude. Please, everyone, suspect all the rest of the science in this article because if something so elemental is wrong, what else is wrong?
The primary scientific source cited in this post would make anything generating a dose of 2 milliGrays an hour (0.002 Gy/hr) intermediate-level radioactive waste (page 5, http://www-pub.iaea.org/MTCD/publications/PDF/Pub815_web.pdf , factoring for w=1 as is the case for beta and gamma at 2 mSv/hr) and require storage in packages ‘with 200 mm reinforced concrete walls’ (page 83, op cit).
This post also does not understand the difference between exposure level (measured in Sv, mSv or uSv) and dose (measured in Gy, mGy or uGy). That’s Health Physics 101, and required for any accurate understanding of the issue. One Sv of field strength results in one Gy of dose as per http://books.google.com/books?id=ZBQXV7glXfQC&pg=PA38&lpg=PA38&dq=background+radiation+Gray&source=bl&ots=ilTeQ7AxnR&sig=4RT9E1nb-7wS3xYRTwwzI0Tnhps&hl=en&ei=2bScTfz4La2N0QG5wJTpAg&sa=X&oi=book_result&ct=result&resnum=27&ved=0CNQBEOgBMBo
That same document (page 76) specifies background radiation in that area as measured in uSv/hr, not mSv/hr, three orders of magnitude less than the author of this blog post mistakenly suggested.
A somewhat more authoritatve source http://www.pbs.org/wgbh/pages/frontline/shows/reaction/interact/facts.html notes the average worldwide background radiation level is 2mSv per YEAR, not per HOUR. 2mSv/yr would result in a dose of 2mGy/yr.
http://rex.nci.nih.gov/NCI_Pub_Interface/raterisk/risks90.html (second graf) confirms an annual dose is on the order of 1-2 mGy/yr, whereas http://www.colorado.edu/EHandS/hpl/RADHandbook/Introduction.html in the first graf of section 2 noted US background level is higher, on the order of 3.6 mSv/yr.
And, a suggestion to the blog post author: Please DO THE MATH.
>> When it comes down to it, nuclear power is “safe”.
You are completely, totally, umambiguously, demonstratively, absolutely wrong.
Not “wrong.”
Wrong.
Does radiation exposure really kill RBCs? They don’t have genetic material to speak of.
Hi John Bartley,
Thank you very much for your thoughtful and thorough post. I’m checking the math now, and I’ll certainly get back to you. I really appreciate your attention to these details.
Hi Mark,
Gamma radiation can kill red blood cells, yes. Elegant explanation:
“Gamma irradiation of red blood cells induces alterations at three different
functional units of the membrane: lipid bilayer, protein components and
cytoskeleton at the membrane surface [3]. In addition, radiation induces shortening
in the lipid fatty acid chains by lipid peroxidation. The production of
hydroperoxides and cross-linkages in the membrane lipids can disorder the upper
region of the bilayer favoring penetration of water and ending by hemolysis.”
Source: “Effect of Gamma Radiation on Some Biophysical Properties of Red Blood Cell Membrane”
Nabila S. Selim, et al.
Blood, Vol 83, No 6 (March 15), 1994: pp 1683-1 689
Also see:
“Ionizing radiation-induced structural modification of human red blood cells”
K. Gwoździński
Radiation and Environmental Biophysics
Volume 30, Number 1, 45-52, DOI: 10.1007/BF01595573
Though, interestingly, gamma rays from Cesium-137 have often been used to irradiate red blood cells in order to prevent transfusion-associated graft-versus-host disease.
See, for example:
“Deformability and viability of irradiated red cells.”
Reverberi R, Govoni M, Verenini M.
Ann Ist Super Sanita. 2007;43(2):176-85.
http://www.ncbi.nlm.nih.gov/pubmed/17634667
I suppose the key is to use the right amount.
Why do people keep saying nuclear power is safe? Will it become true if people keep repeating it? Will Fukushima’s evacuation be cancelled? Will the multi-core meltdown stop in it’s tracks? Yes, there are relatively safer reactor models, yet we are still using these dangerously outdated models worldwide. There is a known issue regarding these particular reactor vessel’s potential to fail during a meltdown. That alone should be evidence that society is not responsible enough to handle power generation from nuclear fission. This situation is already not safe and the crisis has not even reached a conclusion yet!
Umm…safe means harmless. How people can keep saying nuclear energy is “reasonably safe” – especially now – is so beyond me. Is that like being a little bit pregnant?
Look: there’s NO place to put the waste, the spent fuel rods remain hot and dangerous for thousands of years…I mean Hel-Lo. This was a technology that was, in theory, a good idea but was never perfected, brought too soon to market and is NOT safe!
Solar energy is safe. Windmills are safe. OK, a windmill might fall on you and smash your house. But it won’t poison every fish in the ocean; it won’t contaminate cow’s milk halfway around the globe. You know what I’m saying?
So obvious, I could scream.
And, yet another source to demonstrate the bad science in this post: http://www.newscientist.com/blogs/onepercent/2011/04/robots-find-high-radiation-lev.html
The last graf of that story shows normal background radiation at 1/100 (0.01) of a millisievert per hour, which, for beta and gamma, would generate a dose of 0.01 milliGray/hr. That’s 1/200 of the dose of 0.002 Gray/hr you quoted in graf 11 of your story.
How much more proof do you need to correct your error above?
I really do agree with John B. about this post giving the wrong information in so many ways. As a medical physicist I can not even begin to clear all of them up here! Clearly there are misunderstandings about radiation levels and units and the effect of radiation on humans. But the author of this post have not even understood how the small things are related, such as that Leide was not Devairs and Marias daugther. Leides mother did not die.
Even Wikipedia gives a more correct view of the accident. Please read that instead if you want simplified information!
http://en.wikipedia.org/wiki/Goiânia_accident
For a more scientific view please read the International Atomic Energy Agency’s report of the accident.
http://www-pub.iaea.org/MTCD/publications/PDF/Pub815_web.pdf
Please all -do not believe everything you read!
G’Day! Lastwordonnothing,
Thanks for the above, Please mention techniques, tips to avoid contamination while doing experiments and also suggest some tips to do work with maximum efficiency.Please provide links of important articles or videos related to this topic.Thanks in advance.
Keep up the good work