
In the spring of the year 73, thousands of Roman soldiers raided Masada, a fortress on top of a cliff in the Judean Desert. For seven years, the Jews had tried, unsuccessfully, to split from the Roman empire, and Masada was the last holdout. According to the ancient historian Josephus, when the Romans breached Masada’s walls, they found 960 dead bodies of Jewish extremists, called Sicarii, who had killed themselves to avoid the inevitable enslavement. Because of Masada’s remote location and harsh, dry climate, nothing much happened to the site for the next 2,000 years, until archaeologists started digging it up in 1963. They found attack ramps and siege towers (some of the best examples we have, apparently, of Roman war technologies), palaces, cisterns, swimming pools, 27 human skeletons and, deep under the rubble, a handful of seeds.
The seeds were stored at room temperature until 2005, when scientists performed radiocarbon dating and identified them as the famed date palm of Judea. (Psalm 92: “The righteous will flourish like a palm tree…They will still bear fruit in old age, they will stay fresh and green.”) The researchers planted the remaining three seeds. One of them grew. When the results were published, in 2008, the plant, nicknamed Methuselah, was more than three feet tall. By this past November, it was more than six feet tall, and healthy enough to be moved out of quarantine and into a park.
No one knows exactly how the seeds managed to survive so long, but it almost certainly had to do with the extreme dryness of the desert. Methuselah is just one of many examples of organisms that can preserve themselves by shutting down for awhile. In the winter, the wood frog’s heart stops beating and up to 45 percent of its body turns to ice. The tardigrade, a microscopic eight-legged ‘waterbear’, can survive at least 10 years in a dry environment by expelling nearly all of the water from its body.
“Nature is very wise at solving these problems,” says cryobiologist Amir Arav, whose company, Core Dynamics, is based about 85 miles from Masada. For nearly 30 years, Arav has been trying to mimic nature’s preservation feats in the lab. He has frozen rat livers and hearts, and sheep ovaries, and has freeze-dried human sperm, spongey bone, stem cells and blood.
Cryobiology involves complicated physics and chemistry: phase transitions, sublimation, annealing, adsorption isotherms. But Arav says it all boils down to one simple principle. “There is one thing that makes biologic time stop, and that is a solid state.”
Cells are made of mostly water. When water is in a liquid state, molecules move around, biochemical reactions happen, the cell does its work. When you get rid of liquid water — either by drying it out or freezing it — the movement stops, and so does biological activity. “And if you move it back into a liquid state, it will all start back up again,” Arav says.
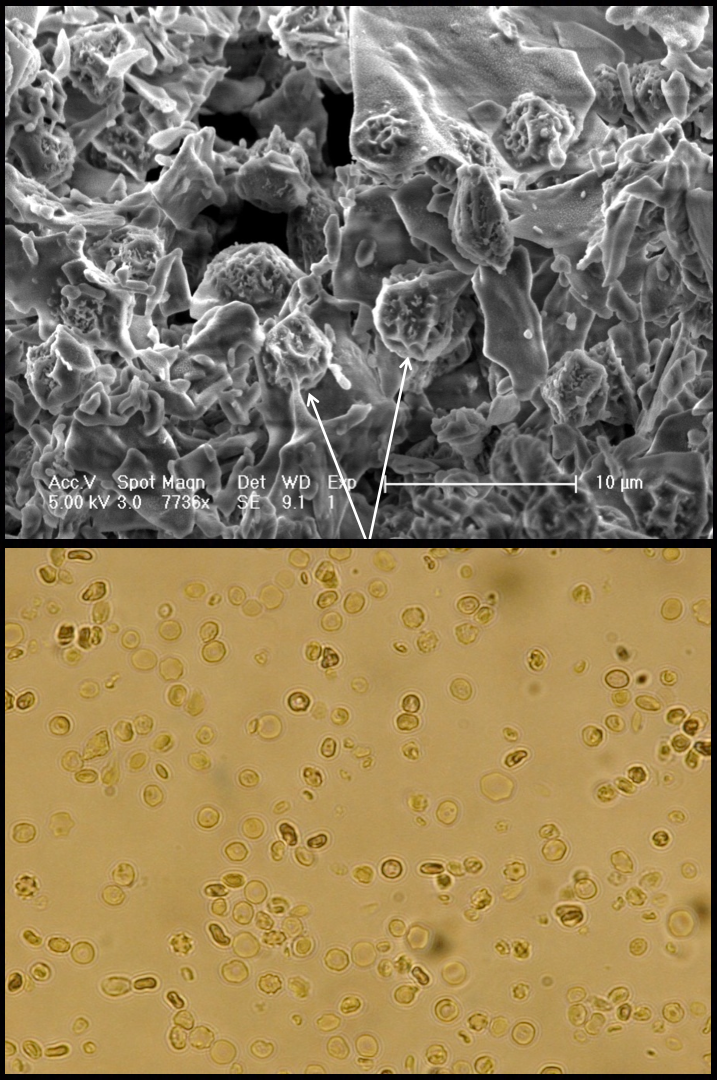
Stopping biological time could be useful for many reasons (some weirder than others), but one of the most practical applications is preserving blood. At room temperature, red blood cells degrade in less than a day; even in a refrigerator, they only last 42 days. This is a big problem for anybody doing medical procedures with limited electricity — in a rural clinic, say, or on the battlefield. Imagine if soldiers could carry around tiny pellets of their own dried blood that, like instant coffee, transform with a few drops of water. Arav’s company has been working with the U.S. Army, in fact, to develop freeze-dried blood. But why freeze the blood first?, I asked Arav. Why not just dry it out, like the date seeds?
Unlike Methuselah and tardigrades, Arav explains, human cells don’t have the specialized proteins and sugars needed to survive the drying process. So you have to freeze them first. That’s not to say that freezing is very good for cells, either: If you do it too slowly, then you get large ice crystals, which damage cells. One way around this is to freeze cells so quickly that the water turns instantly from liquid into a glass state, bypassing crystallization. This process, called vitrification, was “the big breakthrough in the last century of cryobiology,” Arav says.
Most forms of vitrification require the addition of large quantities of cryoprotectants: substances (glycerol, typically) that protect the cells from damage. But these cryoprotectants can also be toxic, especially in the quantities needed for freezing large volumes of cells. Arav’s company has developed another process, called directional freezing, that does not need cryoprotectants. With this method, the researchers push the cells through a temperature gradient — from high temps to low — which allows them to control where the ice crystals form, and keep them out of the cells.
After successful freezing, the scientists dry the cells into powder. So far, Arav has been able to freeze-dry red blood cells with an 88 percent survival rate. He hasn’t done more than a teaspoon at a time, though, so the next step is scaling up the whole process. I asked him when he thought soldiers would be carrying around freeze-dried blood — five years from now? Ten? He laughed, and told me he had just read a book written by a physicist who complained that writers were constantly asking him when we’ll all be able to buy invisibility cloaks. For freeze-dried blood, Arav said, “I know that it’s going to happen. I just can’t tell you exactly when.”
**
Photo of Masada via Flickr; freeze-dried blood cells courtesy of Amir Arav
Wasn’t there also some fossilised bacterium that managed to survive in spore form, going into hibernation way back before Earth went into its snowball/slushball phase?
SIGH. Memory fail. Everything else fail too.
hmmm….this?
http://www.sciencemag.org/content/268/5213/1060.abstract
Ive been waiting 7 years for this company to do something, my investment in there private placement with lew pell and shlomo ben haim looks like a BIG mistake!!!!!!!